Equine Plasma Products
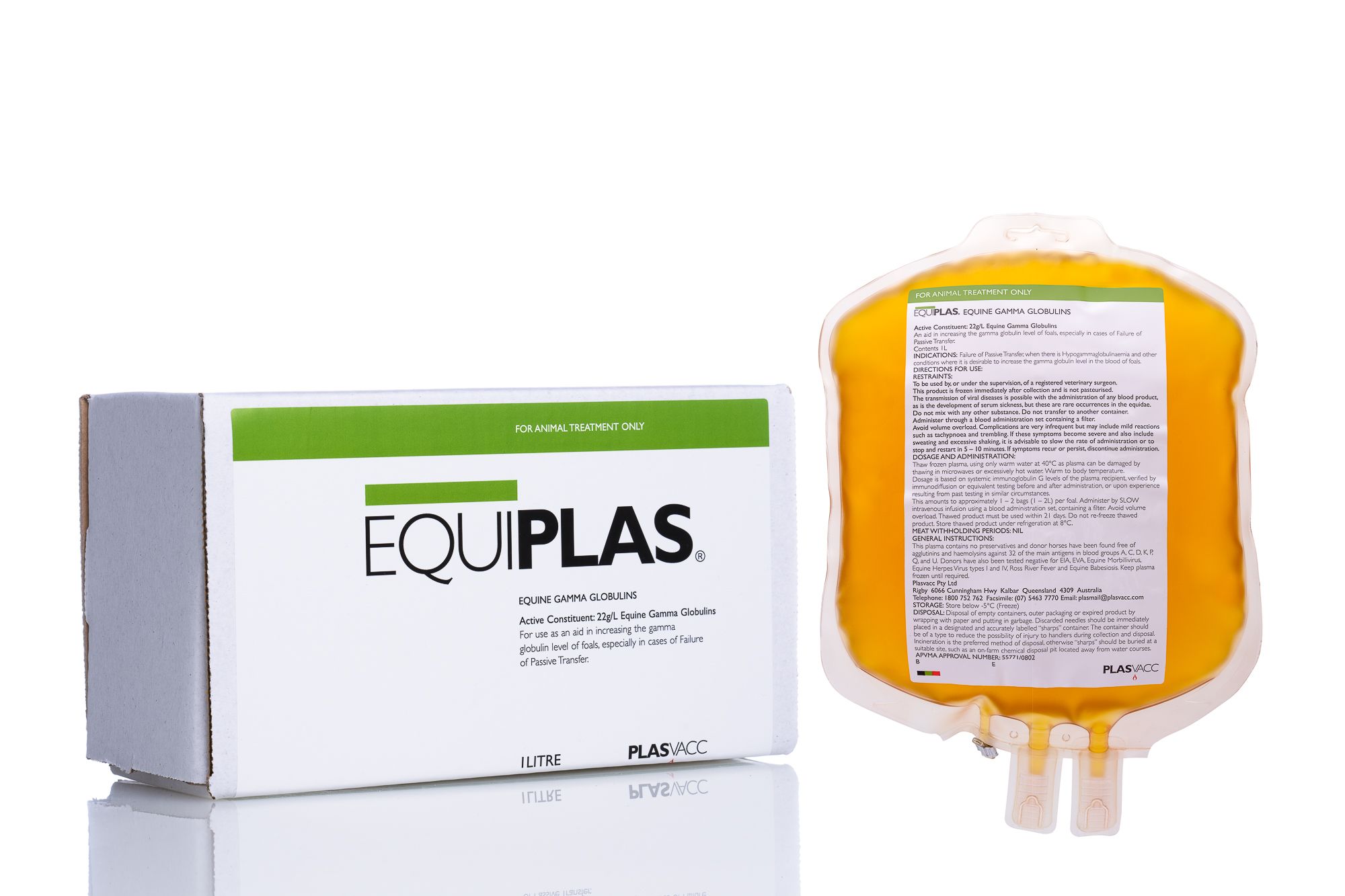
Equiplas®
Equiplas® (Equine IgG) is equine plasma for the treatment of failure of passive transfer (FPT) in the equine neonate. The minimum IgG level is 1800 mg/dl and one 1000-ml bag will increase a healthy foal’s circulating IgG level by about 250 mg/dl. This product is APVMA (Australian Pesticides and Veterinary Medicines Authority) & NZFSA (New Zealand Food Safety Authority) registered.
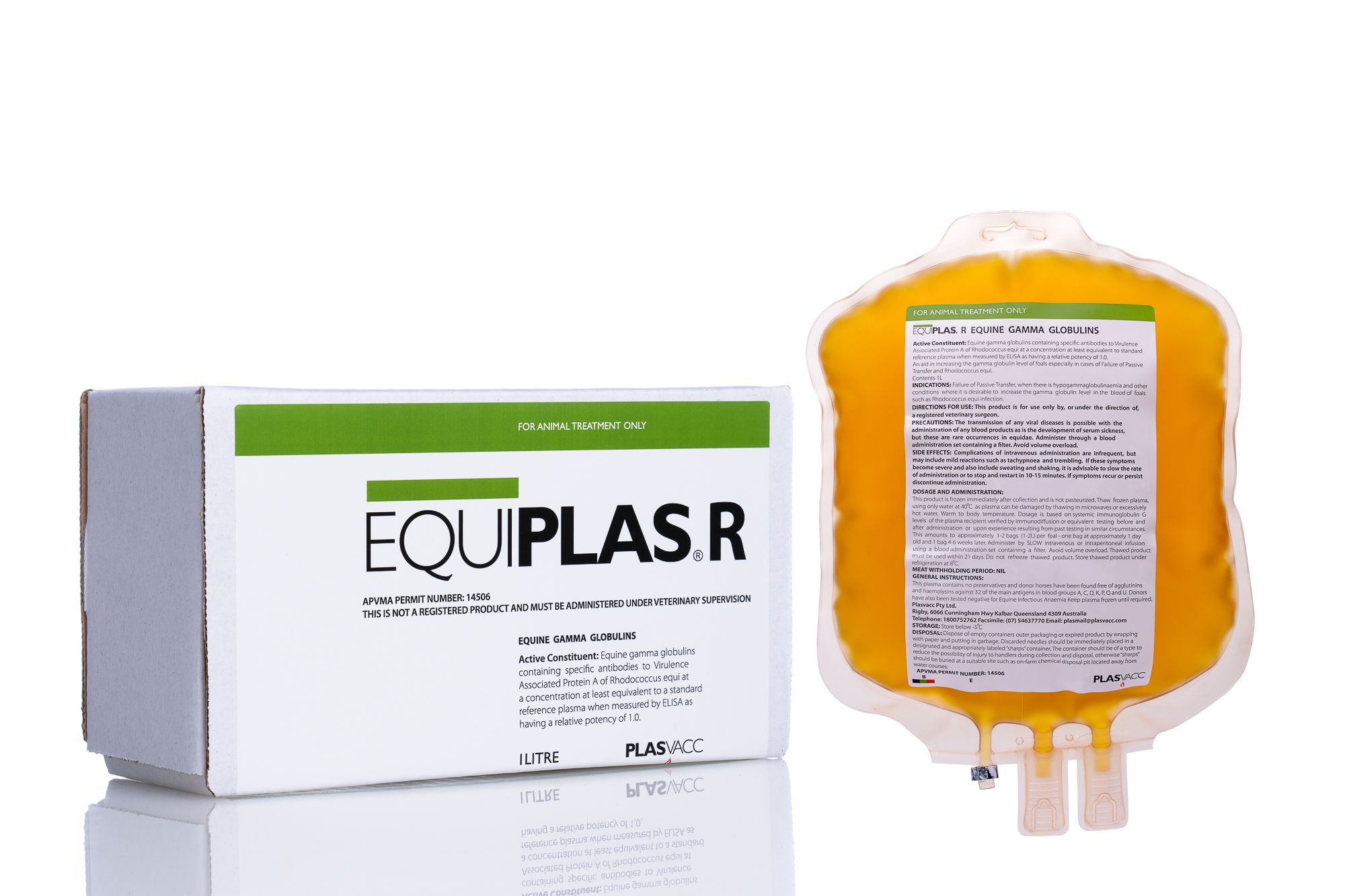
Equiplas® R
Equiplas® R (Equine IgG & Rhodococcus Equi Antibody, Equine Origin) is APVMA (Australian Pesticides and Veterinary Medicines Authority) permit approved for the treatment of failure of passive transfer (FPT) and/or as an aid in the management and control of respiratory disease associated with Rhodococcus equi. The minimum IgG content is 2400 mg/dl. This product is intended for use in foals less than 6 months of age and is supplied in 1000ml bags.
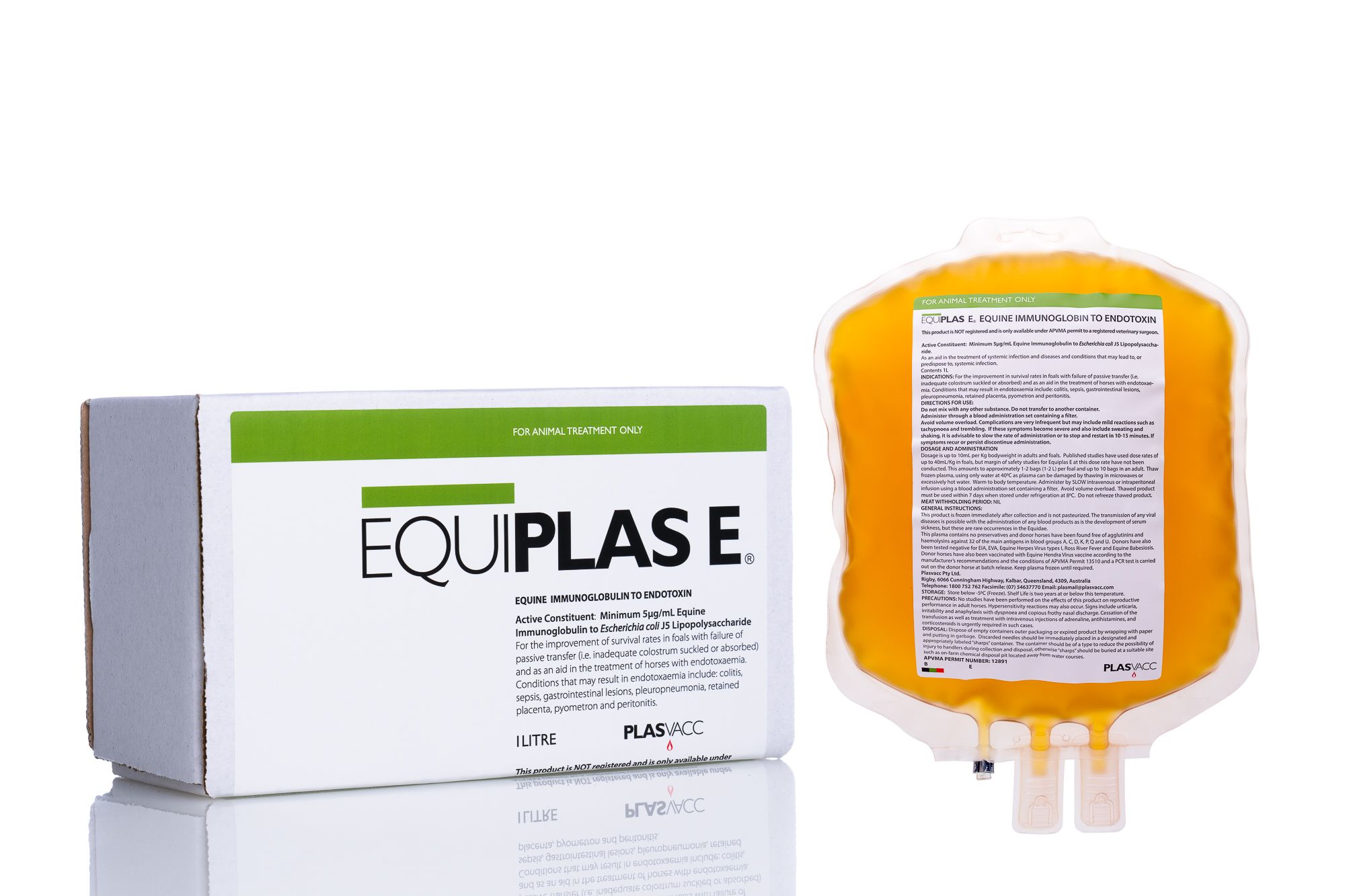
Equiplas® E
Equiplas-E is Plasvacc’s hyperimmunised equine plasma product specifically designed to help treat Endotoxaemia in horses. Rich in specific anti-endotoxin gamma globulins, Equiplas-E is collected from specially hyperimmunised donor horses that reside at the Plasvacc plasma production facility, the only facility of its kind in Australia. Endotoxaemia is a condition whereby bacteria in the process of dying release a protein that is toxic to the host animal. Because it is the toxin that is harmful, rather than the organism itself, which would be susceptible to antibiotics, treatment of this condition is not straightforward. The administration of targeted antibodies is the only specific method of treatment for this condition and such antibodies are contained in Plasvacc’s Equiplas-E product. The development of Equiplas-E represents a significant improvement in the treatment modalities for this condition. Equiplas-E does not replace all present conventional treatments of ETA, rather it complements these treatments and shortens the course of hospitalisation, thereby saving animals that may not normally survive. Equiplas-E is supplied under APVMA (Australian Pesticides and Veterinary Medicines Authority) permit.

